Back
Purpose: A process for the manufacture of multi-particulate minitablets (tablets with a diameter of less than three millimeters and different release profiles) was developed using a simple direct compression with multi-tip tablet tooling and aqueous enteric coating for use in immediate and delayed release clinical studies.
Methods: The materials included the API (Compound A), Lactose Monohydrate, Silicified Microcrystalline Cellulose, Colloidal Silicon Dioxide, magnesium stearate (from vegetable source), non-functional aqueous coating material, aqueous enteric coating material. All materials except the coating materials were blended and compressed using a rotary tablet press. Initial batches (Batches A and B) were manufactured using single tip punches. Later manufacture (Batches C and D) used multi-tip punches (six tips). Coatings were applied using a perforated coating pan with screen inserts.
Results: Due to the small size of the tablets, the smallest available fill cam (6 mm) was used to prevent the recirculation of excess blend. There were no significant differences in the feeder speeds required to maintain tablet weight between the single and multi-tip tooling. In-Process Compression data showed equivalent tablet weight control but better control of tablet hardness with the single tip tooling as assessed by Process Capability (Cpk), see Table 1. Multi-tip tooling doesn’t allow the measurement of individual press force for each tablet, rejection of out-of-spec materials was a concern. However, the calculated Cpk of 1.47 for in-process weights indicates that compression with multi-tip tooling is very capable of producing product (~ 99.994%) within specifications. This was also evidenced by stratified content uniformity data presented in Figure 1. Less variable data was noted for the minitablets produced with multi-tip tooling. The variation of compression forces, via %RSD for each machine recorded compression cycle, was also examined and the multi-tip tooling was found to exhibit a significantly lower level of variation from punch to punch, See Figure 2. This likely results from a combination of higher per tool compression force and the distribution of force across the six punch tips. During process development studies, chipped and broken tablets were observed. Investigation determined that the ejection height of the tooling was the root cause; punch tips extended a few thousandths of an inch above the surface of the die. The multi-tip tools have six tips arranged in a circle. This resulted in the leading tablets being successfully removed from the lower punch tip but were caught between the knock off bar and the trailing tooling tip, resulting in chipped and broken tablets. Ejection cam shims were removed to lower the height. The press was turned manually then at increasing speeds to verify correct ejection. After adjustment, no broken tablets were further noted. Minitablet cores were seal coated with a non-functional aqueous coating material to a weight gain of 5%. Seal coated cores were then coated to a weight gain of 40% with an aqueous enteric coating material. Lower levels of enteric coating had previously been tested but were not sufficient to protect the minitablet core from acid intrusion during the simulated gastric stage of the dissolution testing. Minitablet non-functional and enteric coating were successfully performed by initially using an elevated tablet bed temperature and gradually reducing it to target while ensuring the spray rate was suitable. This approach resulted in some static based sticking within the coating pan, but it greatly reduced the chance of over-wetting the tablet bed. Quality inspections on the final coated product yielded no defects of any nature for the critical, major and minor categories. Coated minitablet dissolution data readily met the acceptance criteria.
Conclusion: While the manufacture of minitablets is fundamentally no different than that of normal sized tablets, widespread implementation has been impeded due to technical constraints. Multi-tip tooling was successfully implemented in Minitablet manufacture of Compound A based on a combination of low compression forces, superior material flow characteristics and equipment modifications in addition to statistical analysis of process capability. Perforated pan coating of minitablets presented challenges related to the low weight and high surface area of the minitablet bed as well as the nature of delayed release coating materials. The low weight of minitablets makes them susceptible to static, resulting in the minitablets sticking to each other and subsequently twinning during coating. This effect can be exacerbated during enteric coating, which typically occurs at lower bed temperatures and can result in over-wetting which leads to additional twinning or sticking to the interior surfaces of the coating pan sufficient to cause a batch failure due to low yields or too many appearance defects. Precise control of the thermodynamic conditions in coating pan helped offset the issues discussed above and achieve a successful product meeting all the critical quality attributes.
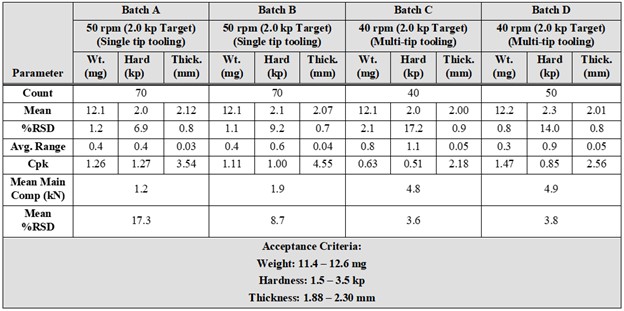 Table 1: In process Compression Data and Process Capability Analysis for Minitablets
Table 1: In process Compression Data and Process Capability Analysis for Minitablets
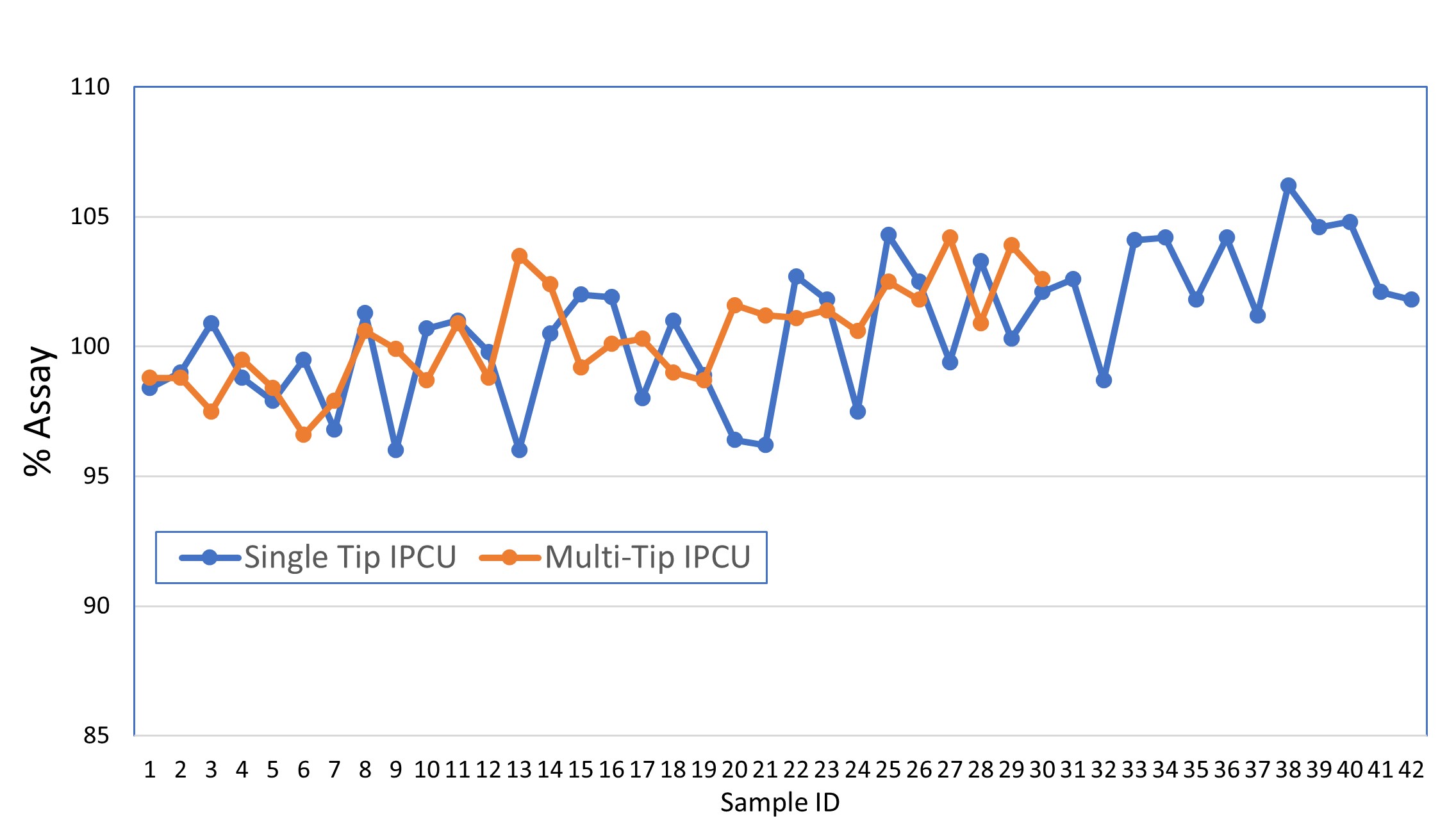 Figure 1: Comparison of in-process content uniformity for minitablets: Single tip vs Multi-tip tooling
Figure 1: Comparison of in-process content uniformity for minitablets: Single tip vs Multi-tip tooling
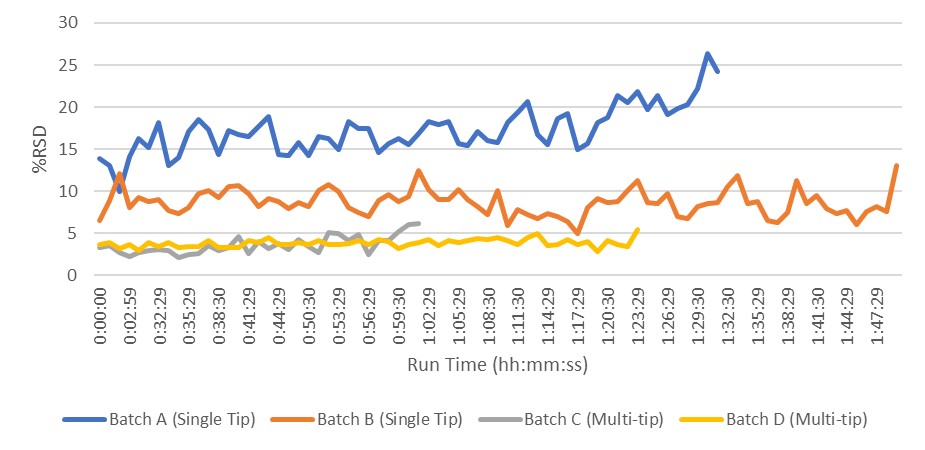 Figure 2: Compression Force %RSD comparison for single tip vs multi-tip batches
Figure 2: Compression Force %RSD comparison for single tip vs multi-tip batches
Formulation and Delivery - Chemical
Category: Late Breaking Poster Abstract
(W1130-03-21) Perforated Pan Coating of Directly Compressed Minitablets with the Use of Multi-tip Tooling to Facilitate Multi-particulate Drug Delivery: Challenges and Outcomes
Wednesday, October 25, 2023
11:30 AM – 12:30 PM ET
- JV
Jason Vogt, B.S.
Alcami Corporation
Wilmington, North Carolina, United States - JV
Jason Vogt, B.S.
Alcami Corporation
Wilmington, North Carolina, United States
Presenting Author(s)
Main Author(s)
Purpose: A process for the manufacture of multi-particulate minitablets (tablets with a diameter of less than three millimeters and different release profiles) was developed using a simple direct compression with multi-tip tablet tooling and aqueous enteric coating for use in immediate and delayed release clinical studies.
Methods: The materials included the API (Compound A), Lactose Monohydrate, Silicified Microcrystalline Cellulose, Colloidal Silicon Dioxide, magnesium stearate (from vegetable source), non-functional aqueous coating material, aqueous enteric coating material. All materials except the coating materials were blended and compressed using a rotary tablet press. Initial batches (Batches A and B) were manufactured using single tip punches. Later manufacture (Batches C and D) used multi-tip punches (six tips). Coatings were applied using a perforated coating pan with screen inserts.
Results: Due to the small size of the tablets, the smallest available fill cam (6 mm) was used to prevent the recirculation of excess blend. There were no significant differences in the feeder speeds required to maintain tablet weight between the single and multi-tip tooling. In-Process Compression data showed equivalent tablet weight control but better control of tablet hardness with the single tip tooling as assessed by Process Capability (Cpk), see Table 1. Multi-tip tooling doesn’t allow the measurement of individual press force for each tablet, rejection of out-of-spec materials was a concern. However, the calculated Cpk of 1.47 for in-process weights indicates that compression with multi-tip tooling is very capable of producing product (~ 99.994%) within specifications. This was also evidenced by stratified content uniformity data presented in Figure 1. Less variable data was noted for the minitablets produced with multi-tip tooling. The variation of compression forces, via %RSD for each machine recorded compression cycle, was also examined and the multi-tip tooling was found to exhibit a significantly lower level of variation from punch to punch, See Figure 2. This likely results from a combination of higher per tool compression force and the distribution of force across the six punch tips. During process development studies, chipped and broken tablets were observed. Investigation determined that the ejection height of the tooling was the root cause; punch tips extended a few thousandths of an inch above the surface of the die. The multi-tip tools have six tips arranged in a circle. This resulted in the leading tablets being successfully removed from the lower punch tip but were caught between the knock off bar and the trailing tooling tip, resulting in chipped and broken tablets. Ejection cam shims were removed to lower the height. The press was turned manually then at increasing speeds to verify correct ejection. After adjustment, no broken tablets were further noted. Minitablet cores were seal coated with a non-functional aqueous coating material to a weight gain of 5%. Seal coated cores were then coated to a weight gain of 40% with an aqueous enteric coating material. Lower levels of enteric coating had previously been tested but were not sufficient to protect the minitablet core from acid intrusion during the simulated gastric stage of the dissolution testing. Minitablet non-functional and enteric coating were successfully performed by initially using an elevated tablet bed temperature and gradually reducing it to target while ensuring the spray rate was suitable. This approach resulted in some static based sticking within the coating pan, but it greatly reduced the chance of over-wetting the tablet bed. Quality inspections on the final coated product yielded no defects of any nature for the critical, major and minor categories. Coated minitablet dissolution data readily met the acceptance criteria.
Conclusion: While the manufacture of minitablets is fundamentally no different than that of normal sized tablets, widespread implementation has been impeded due to technical constraints. Multi-tip tooling was successfully implemented in Minitablet manufacture of Compound A based on a combination of low compression forces, superior material flow characteristics and equipment modifications in addition to statistical analysis of process capability. Perforated pan coating of minitablets presented challenges related to the low weight and high surface area of the minitablet bed as well as the nature of delayed release coating materials. The low weight of minitablets makes them susceptible to static, resulting in the minitablets sticking to each other and subsequently twinning during coating. This effect can be exacerbated during enteric coating, which typically occurs at lower bed temperatures and can result in over-wetting which leads to additional twinning or sticking to the interior surfaces of the coating pan sufficient to cause a batch failure due to low yields or too many appearance defects. Precise control of the thermodynamic conditions in coating pan helped offset the issues discussed above and achieve a successful product meeting all the critical quality attributes.
 Table 1: In process Compression Data and Process Capability Analysis for Minitablets
Table 1: In process Compression Data and Process Capability Analysis for Minitablets Figure 1: Comparison of in-process content uniformity for minitablets: Single tip vs Multi-tip tooling
Figure 1: Comparison of in-process content uniformity for minitablets: Single tip vs Multi-tip tooling Figure 2: Compression Force %RSD comparison for single tip vs multi-tip batches
Figure 2: Compression Force %RSD comparison for single tip vs multi-tip batches