Formulation and Delivery - Chemical
Category: Late Breaking Poster Abstract
(M0930-03-20) Optimization of Roller Compaction Process Parameters for a Poorly Soluble Drug Using Quality by Design Tool
Monday, October 23, 2023
9:30 AM - 10:30 AM ET
- KP
Kuntal Parikh, M.S. (he/him/his)
Patheon, part of Thermo Fisher Scientific
Brampton, Ontario, Canada - KP
Kuntal Parikh, M.S. (he/him/his)
Patheon, part of Thermo Fisher Scientific
Brampton, Ontario, Canada - MA
Marwa Abdalla, M.Sc. (she/her/hers)
Patheon, part of Thermo Fisher Scientific
Mississuga, Ontario, Canada - LK
Lalji Kathiria
Patheon, part of Thermo Fisher Scientific
Mississauga, Ontario, Canada - MB
Milan Babu
Patheon, part of Thermo Fisher Scientific
Mississauga, Ontario, Canada - RS
Rupninder Sandhu
Patheon, part of Thermo Fisher Scientific
Mississauga, Ontario, Canada - BG
Barry Gujral
Patheon, part of Thermo Fisher Scientific
Mississauga, Ontario, Canada
Presenting Author(s)
Main Author(s)
Co-Author(s)
Purpose: To optimize roller compaction process parameters for an immediate release tablet of a poorly soluble drug using Quality by design (QbD) strategy. QbD Tools Used: Minitab® 17
Methods: Roller compaction process was developed at small scale using Gerteis Mini-Pactor® followed by compression and coating. The process parameters and the ranges were identified based on initial prototype manufacturing trials and manufacturing process risk assessment. A 23-1 fractional factorial Design of Experiments (DoE) was conducted for independent variables viz. roll gap, roll force and granulator screen size as the process parameters for roller compaction (Attachment 1). Total of 7 roller compaction trials including 3 centre-points were designed and manufactured. To keep a tablet solid fraction constant, each final blend was compressed to the same tablet thickness. The effect of the input variables on the response factor including ribbon solid fraction, amount of fines, tablet tensile strength, % dissolved in 30 minutes, friability and disintegration time were evaluated (Attachment 1). For each response factor, analysis of variance (ANOVA) testing and multiple regression analysis was performed to determine the optimum regression equations describing the effects of the input variables on the modeled response. Regression analysis was performed using Minitab®17 to obtain normal and residual plots.
Results: During manufacturing of the DoE batches, technical challenges were not observed. The ribbon solid fraction was in the range of 0.58 to 0.70. These results indicate that the solid fraction of the ribbons was impacted roll force and roll gap within the studied range. The DoE summary for model robustness indicated that for the ribbon solid fraction, the R2 value is 96.49% and R2 adjusted is 94.74%. The DoE summary indicated poor model for the amount of fines and tensile strength as R2 value are 56.68% and 54.62%, respectively and, R2 adjusted are 48.02% and 45.55%, respectively. No good fit statistical model was found to relate changes in % dissolved in 30 minutes, friability, and disintegration time as R2 adjusted is below 20.0%. Based on the regression equation, combination of roll gap and roll force were simulated per attachment 2 which can target 0.6 solid fraction. A confirmatory batch has been planned to confirm the model for ribbon solid fraction at scale.
Conclusion: Based on mathematical and mechanistic modeling, it is concluded that roll gap and roll force are Critical Process Parameters (CPPs) and they have impact on ribbon solid fraction. The confirmatory batch is planned to further validate the mathematical/mechanistic modeling.
.jpg) Table 1: Summary of input and output factors
Table 1: Summary of input and output factors Figure 1: 2-level factorial design matrix
Figure 1: 2-level factorial design matrix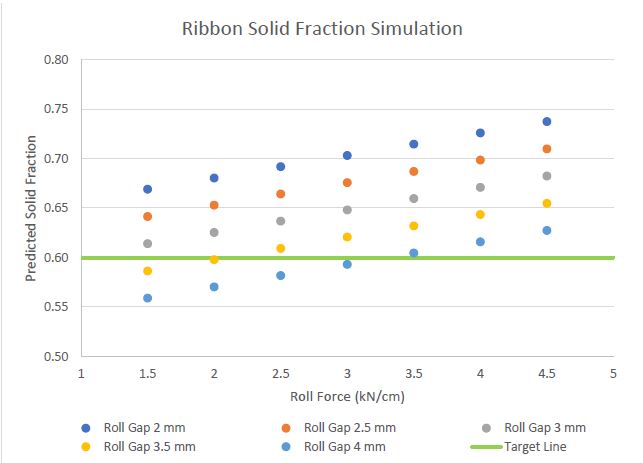 Figure 2: Predicted ribbon solid fraction from model simulation
Figure 2: Predicted ribbon solid fraction from model simulation