Formulation and Delivery - Chemical
Category: Late Breaking Poster Abstract
(M1130-03-18) Toward IVIVC for ASDs by Convolution of First Derivative Permeation Profiles
- JJ
Jacob Rune Jørgensen, Ph.D. (he/him/his)
Bioneer A/S
Copenhagen, Hovedstaden, Denmark - JJ
Jacob Rune Jørgensen, Ph.D. (he/him/his)
Bioneer A/S
Copenhagen, Hovedstaden, Denmark - WM
Wolfgang Mohr, Ph.D.
Losan Pharma
Neuenburg, Baden-Wurttemberg, Germany 
Matthias Rischer, Ph.D.
Director Drug Delivery & Innovation Projects
Losan Pharma
Neuenburg, Baden-Wurttemberg, Germany- AS
Andreas Sauer, Ph.D. (he/him/his)
SE Tylose GmbH & Co. KG
Wiesbaden, Hessen, Germany - SM
Shilpa Mistry, Ph.D.
Harke Pharma
Urmston, Manchester, England, United Kingdom - TR
Thomas Rades, Prof. Dr. Dr. h.c. (he/him/his)
University of Copenhagen
Copenhagen, Hovedstaden, Denmark - AM
Anette Müllertz, Ph.D. (she/her/hers)
University of Copenhagen
Copenhagen, Hovedstaden, Denmark
Presenting Author(s)
Main Author(s)
Co-Author(s)
Purpose: In vitro-in vivo correlation (IVIVC) is defined by the U.S. Food and Drug Administration as “a predictive mathematical model describing the relationship between the in vitro property of a dosage form and an in vivo response”. The highest level of correlation (Level A) further represents a point-to-point relationship between in vitro dissolution and the in vivo pharmacokinetic (PK) profile. To achieve such correlation, careful consideration of the in vitro setup is necessary to ensure predictive in vivo performance. However, for enabling formulations, e.g., amorphous solid dispersions, establishing IVIVC can be further complicated due to the challenge of distinguishing molecularly dissolved drug from colloidal drug, e.g. polymer-bound drug, drug solubilized in micelles or included into cyclodextrin [1]. The aim of the present work was to convert in vitro drug permeation data into dissolution profiles for molecularly dissolved drug and further convolute the dissolution profiles to fit in vivo PK data of ASDs.
Methods: Raw data was obtained from a recently published study on dissolution-permeation in vitro tests of four binary ASDs of efavirenz (EFV) and crystalline EFV which achieved an in vitro-in vivo relationship (IVIVR) with R2 = 0.97, yet without an IVIVC point-to-point correlation. A significantly improved relationship between in vitro drug permeation and in vivo absorption was realized by modifying the permeation barrier with the addition of a hydrophilic filter to obstruct any potential contact between dispersed formulation particles in the in vitro donor compartment and the phospholipid-based membrane barrier (PAMPA) [2]. The present work involved curve fitting of the in vitro permeation profiles by polynomial regression analysis and further conversion to profiles for molecularly dissolved drug by first derivative analysis. The profiles for molecularly dissolved drug were further converted to predicted PK profiles by a convolution-like approach using an EFV elimination half-life of 1 h and a bioavailability factor of 100%. Due to the arbitrary units of the derived profiles for molecularly dissolved drug, the input for the volume of distribution was fixed at an arbitrary value that matched the Cmax of the best in vivo performing ASDs.
Results: The first derivative profiles mimic the dissolution rates of EFV excluding non-absorbable species. Convolution of the first derivative profiles resulted in predicted PK profiles of the four ASDs closely resembling the in vivo PK data (Figure 1). However, a two-fold higher Cmax was predicted for the crystalline drug compared to the in vivo PK data with the present convolution parameters. Thus, the present data treatment suggests that the crystalline drug overperforms relative to the four ASDs in the in vitro dissolution-permeation setup that was previously established with an IVIVR with R2 = 0.97 [2].
Conclusion: The principle of using the first derivatives of the in vitro permeation profiles is a promising tool for deriving the release of molecularly dissolved drug towards establishing IVIVC from ASDs. The method revealed either an overprediction of the crystalline formulation or an underprediction of the four ASDs by the present in vitro setup. While the current data did not lead to a perfect level A IVIVC, the principle can likely be used for further in vitro setup optimization towards eventually establishing true point-to-point IVIVC of enabling formulations.
References: [1] S.T. Buckley, K.J. Frank, G. Fricker, M. Brandl, Biopharmaceutical classification of poorly soluble drugs with respect to “enabling formulations,” European Journal of Pharmaceutical Sciences. 50 (2013) 8–16.
[2] J.R. Jørgensen, W. Mohr, M. Rischer, A. Sauer, S. Mistry, T. Rades, A. Müllertz, In vitro-in vivo relationship for amorphous solid dispersions using a double membrane dissolution-permeation setup, European Journal of Pharmaceutics and Biopharmaceutics. 188 (2023) 26–32.
Acknowledgements: The animal experiments were carried out in agreement with the Danish law on animal experiments as approved by the Danish Animal Experiments Inspectorate in accordance with the EU directive 2010/63/EU under license number 2019–15-0201–00262.
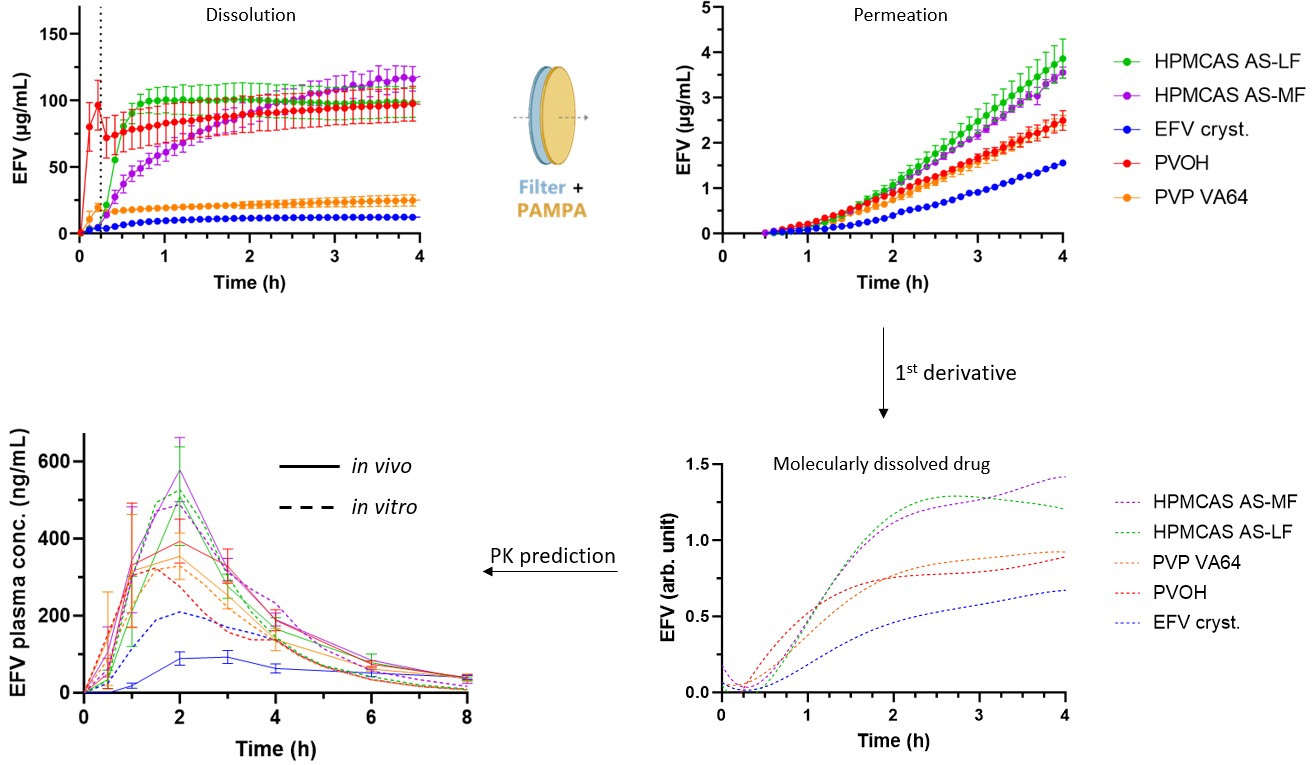 Figure 1. Overview of the data treatment starting with the apparent in vitro dissolution of crystalline EFV and four binary EFV ASDs with HPMCAS AS-LF, HPMCAS AS-MF, PVOH or PVP VA64 as polymer matrix. First derivative analysis of the permeation profiles leads to the profiles of molecularly dissolved EFV which are further converted to predicted PK profiles (dashed lines) plotted together with the actual in vivo PK data (solid lines).
Figure 1. Overview of the data treatment starting with the apparent in vitro dissolution of crystalline EFV and four binary EFV ASDs with HPMCAS AS-LF, HPMCAS AS-MF, PVOH or PVP VA64 as polymer matrix. First derivative analysis of the permeation profiles leads to the profiles of molecularly dissolved EFV which are further converted to predicted PK profiles (dashed lines) plotted together with the actual in vivo PK data (solid lines).